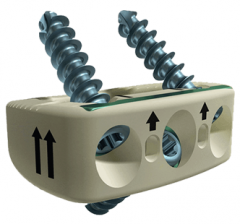
CASCADIA™ AN 3D Interbody System
Category:
Degenerative, Interbody Devices
The CASCADIA AN 3D Interbody System includes a full range of implant sizes carefully designed to accommodate the vertebral anatomy. This system is used in conjunction with instrumentation that allows for both direct implant insertion and in-situ rotation. Lamellar 3D Titanium Technology incorporates 500 um longitudinal channels throughout the implant, which in conjunction with transverse windows, create an interconnected lattice designed to allow for bony integration. [1]






























 Loading...
Loading...