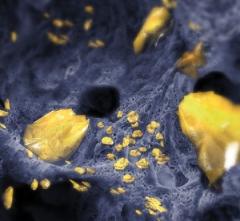
BIO4 possesses all four characteristics involved in bone repair and regeneration: osteoconductive, osteoinductive, osteogenic, and angiogenic.1-3 It is an alternative to autograft that minimizes the potential for harvest site comorbidities. BIO4:
-
Is lot tested for the presence of VEGF (vascular endothelial growth factor)
-
Is lot tested for ≥ 70% cell viability post-thaw
-
Contains ≥ 600,000 cells/cc (endogenous bone forming cells including mesenchymal stem cells)
-
Is ready to use out of the package; no decanting is required, and thaws in 15 minutes
-
Has differentiated handling compared to competition
-
Is non-immunogenic
-
Is available in 1, 2.5, 5 and 10cc sizes
BIO4 is manufactured by Osiris Therapeutics, Inc.
References:
-
Data on file at Osiris Therapeutics, Inc.
-
Roberts and Rosenbaum, “Bone grafts, bone substitutes and orthobiologics,” Organogenesis (2012).
-
Bourke et al., “Vascular Endothelial Growth Factor (VEGF) in Human Peristeum - Normal Expression and Response to Fracture,” Journal of Bone & Joint Surgery, British Volume (2003).



























 Loading...
Loading...